This month's SGS proderm story gets to grips with the subject of skin sensitization, the HRIPT and related regulations. To help us navigate this slightly confusing landscape we welcome Professor Dr. Klaus-Peter Wilhelm.
- Kategorie
- Datum
- Teilen
Prof. Wilhelm, thank you for joining us! To start with, let's set the scene with an obvious question, what is a HRIPT?
Prof. Wilhelm: It is a patch test, to see if a product has the potential to cause a contact sensitization (contact allergy). I must stress it is the potential we are looking for. For ethical reasons we have to assume that the product is not sensitizing. We need to have some certainty that such a potential does not exist, but we can never be sure, so we need the test before the product goes in to market.
Another obvious question: What is the difference between a skin irritation reaction and a skin sensitization reaction?
Prof. Wilhelm: Most intolerance reactions of the skin fall into the first category, i.e. irritation. Skin irritation is something that would affect any person if the exposure conditions (i.e. the amount and concentration of the irritant as well as exposure time) are sufficiently high enough. Some people may have a lower threshold, others a higher one, but if the concentration is high enough, anyone would get irritant dermatitis. This is not the case with sensitization / contact allergy. Here the individual must have a genetic predisposition. Take for example a very common sensitizer, nickel. It is very common, so one can come into contact with nickel through jewelry or by other means, and around 10- 20 % of the general population will become sensitized, which is a high percentage! But nonetheless 80 % will not get sensitized, even though many of them have had the similar exposure, as they do not have the genetic predisposition. This difference between irritation and sensitization is important to know because it has a big impact on the number of subjects having to be included in such a test for exclusion of sensitization. For irritation tests we usually have a panel size of 30 subjects. For a HRIPT, which is a test to exclude sensitization, the standard panel size is 200!
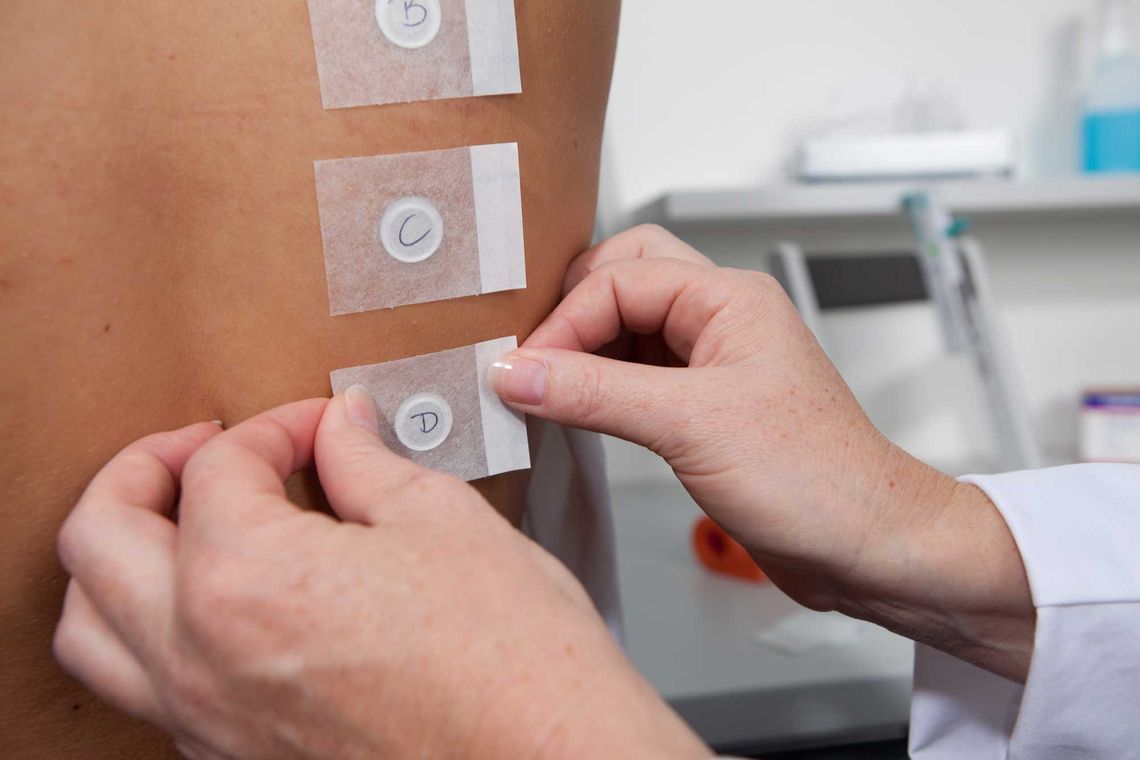
So, what do our study participants undertake when they volunteer for such a study, could you describe the HRIPT procedure?
Prof. Wilhelm: The design has been around for decades, it was first described in the 1950's. It has undergone only a few modifications since then, but the design used today has been used routinely for the last three decades. It’s a 6-week duration study with three phases; the 1st phase is the Induction phase. It extends over 3 weeks, with exposures three times a week. Occlusive patches containing the test substances are usually applied to the upper arm for 48 or 72 hours. This induction phase is so called because sensitization not only requires a genetic predisposition, but also does not occur immediately and only after repeated exposure. This is followed by a 1-week rest phase (the 2nd phase), during which nothing is done. And after that, comes the 3rd and so called challenge phase. In this phase we apply the same material to naïve skin, so the skin of the opposite arm (the one not previously exposed), to see if there is a systemic immune response to the applied material. Again the application is done for 48 hours. Afterwards we perform assessments 15 minutes, 24 hours, 48 hours, 72 hours and sometimes even 96 hours after removal of the test material. The evaluation and interpretation of results is done by a dermatologist who has experience in evaluating allergic diagnostic patch tests. Many products sometimes cause some dermatitis when applied occlusively to the skin, and we need to determine if the reaction we see in the challenge phase is sensitization or just irritation.
How does one do that, recognize the difference between an irritation and a sensitization reaction?
Prof. Wilhelm: One criterion for contact allergy is that the reaction increases after we remove the test material; this is called a crescendo reaction. Thus, the reaction after 48 hours is stronger than after 15 minutes and 24 hours (as evaluated by a dermatologist using a rating system: the higher the rating the stronger the skin reaction). Whereas irritation usually subsides with time, sensitization increases with time. Another criterion which is indicative of contact sensitization is that the reaction in the challenge phase is significantly higher than during the induction phase, so we can see that something is going on in that individual. These two criteria need to be considered by someone who has quite some experience in interpretation of patch tests, especially HRIPTs.

This procedure which you have just described, does it follow an official guideline – is there such a thing?
Not from the regulatory standpoint, at least not exactly. First of all, the procedure I just described has been widely published in the dermatology literature and, funnily enough there is no explicit general guidance for the development of such a testing procedure for topical products in clinical development before obtaining market authorization. However, there is indirect guidance related to specific products. For example, the first guidance issued by the FDA for transdermal delivery systems, specifies the procedure I described. There are also several other guidelines for specific products but not for all. Therefore it is common practice to just use one of these product specific guidelines as a guide of reference for any other topical drug development.
It is possible that a HRIPT may lead to sensitization of subjects, how is this viewed ethically?
This point is raised quite often. I have a clear position on this. Let's look at the situation: Yes, there is a potential possibility that some people will be sensitized even if we start with the assumption that the product is not sensitizing. And yes, this is of ethical concern as even as the visible reaction subsides within a few days, the sensitization might persist for a long time, potentially life-long. But what would be the alternative? Not to perform the test and then go to market, only to discover that the same percentage of the general population becomes sensitized? How can this be ethical? Some people are against the HRIPT, but recommend a 21 day cumulative irritation test instead, looking only at irritation. In my view, that is hypocritical because if you only apply products for 3 weeks and then stop, you still have induced the same rate of sensitization as would have occurred in the HRIPT but you do not detect it only because you didn't look for it. Not only this is hypocritical, but it also puts the affected individual at a great disadvantage because they do not know that they have been sensitized. So, if a sensitization reaction is discovered in a study, then of course it is investigated and the cause is determined, and the subject informed that they have a sensitization to molecule X. That way they could avoid this substance and any related substances because sensitization is potentially life long. This is rightfully of ethical concern and has to be considered when weighing the risks vs. benefits of this study.
What risk assessment must be conducted prior to a HRIPT?
Usually unless the formulation is well known and all the ingredients have extensive clinical data relating to them, as a prerequisite to initiating a clinical study the sponsor must carry out at least an LLNA (local lymph node assay) as well as other preclinical studies depending on the product.
Actually that leads to a question which I had at the back of my mind: at what stage of product development is an HRIPT usually carried out? Because as you described, conducting this type of study is quite investment intensive. So is it done at the end of development, just before regulatory approval for entry into the market or earlier? And in your experience, is it ever done with individual compounds, such as new ingredients for example, or just the final product formulation?
To answer your second question first: single ingredients are very rarely considered for an HRIPT, usually it is final formulations. Your first question is a very important one, but actually there is no unanimous answer (smiles)! You may already know that we held a webinar two years ago on this topic of dermal safety studies, and actually repeated it last year because of great interest in this topic. And there is still the question of when is the best time to do a HRIPT if you are planning a clinical trial anyhow; should it be done fairly early, even before proof of concept; after proof of concept but before the clinical study; or even at the end, when it is a regulatory requirement and you have successfully completed your clinical study. So actually in our webinar I posed the same question regarding the timing in the clinical development plan to the audience who had performed or plan to perform these studies for their products: The answer was that it rarely happened before proof of concept (and only in exceptional cases); and it was more or less split 50:50 between: after proof of concept before the clinical study; or after the clinical study. I think it is always a case-by-case decision, so how well do you know your formula, how sure are you that this test is just a regulatory requirement and nothing else depends on it, or do you really want to be sure before going into clinical studies.
So, what should sponsors do if they are not sure whether a HRIPT is required?
They can come to us and we can advise them with our experience. We also conduct workshops and deal with other aspects of product development. Usually, in the end, it comes down to the recommendation to seek regulatory advice, create a clinical development plan, write up the synopsis, and then go to the regulatory authority to get their opinion.
Thank you Professor Wilhelm for a very interesting and enlightening discussion!
Interviewee

Prof. Dr. Klaus-Peter Wilhelm
Former General Manager and Medical Director




