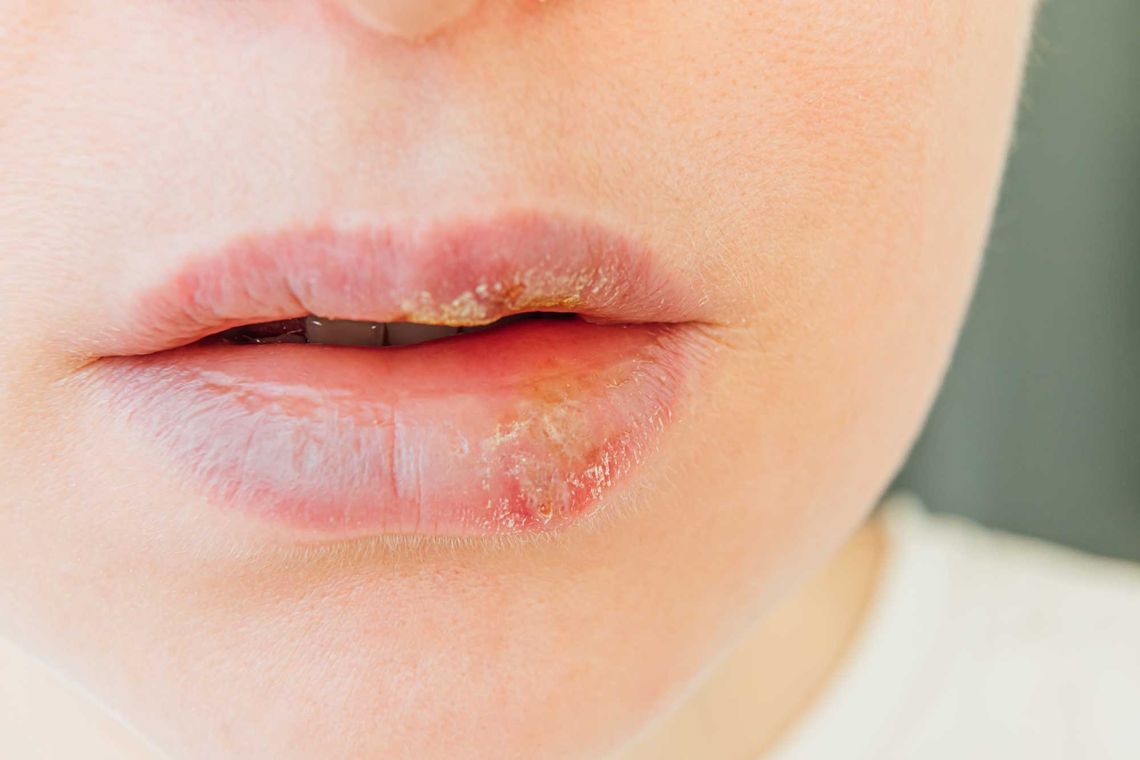
We caught up with Stephan Bielfeldt, our Vice President Science and Consultancy, to talk about Herpes in clinical research – and how we at proderm have all the tools at our fingertips to make these lips talk!
- Kategorie
- Datum
- Teilen

Stephan Bielfeldt
Senior Expert Science & Innovation
Interview with Stephan Bielfeldt
Hello Stephan! Today I would like to probe you a little bit about clinical trials on Herpes simplex! Considering the type of studies we conduct at proderm, what are the key skills we have at proderm, when it comes to clinical trials with herpes patients?
Yes, at proderm we are very adept at proof-of -concept studies, which are primarily about seeing whether a new product can be successful in therapy. To do this, of course, you have to find many patients with acute herpes simplex, aka a cold sore. The standard procedure is to screen a large number of people who have recurrent and frequent outbreaks of herpes simplex on their lips. When they reach the so-called prodromal stage, i.e. when they notice that their lips are tingling, then they should come to the test center very quickly and are then only included if it is actually a herpes cold sore.
Yes, and is that problematic? Herpes is relatively widespread.
Well, yes, but that's more of a random thing - you can have the misfortune that only one in 10 comes to the study site in this defined time period and you end up starting the study with all the patients participating widely scattered and isolated over many days and weeks. If you want to test a new therapy quickly and compactly, then you don't want to do a very long clinical study with tens of tens of weeks. You basically want to specifically trigger herpes and include the patients in a controlled manner over the span of a few days.
So by triggering Herpes in patients, we can skip the tedious process of waiting for the patients to have a cold sore attack?
Exactly! This is particularly useful for early phase studies, where the goal is to do monocentric studies with a simple design and a comparatively short timeframe! At proderm, we can set up a Herpes trial without involving 20 centers and x subjects; you can save that for phase 3!
And what do we use to trigger the cold sore?
On the basis of older American publications we use UV radiation in order to trigger the cold sore. It then usually develops within one or two days after radiation
Why is this not the standard procedure?
Well, in America this method has been used sporadically, in Europe it has never been used before. That is why we wanted to establish this method in a clinical study and introduce it to the European scientific community after careful examination! Unfortunately, the American publications were done with rather outdated models of UV lamps that needed a lot of optimizing for safe and reproducible use on patients. That is why we, together with a sponsor who wanted to test a cold sore product, planned and carried out an exploratory study.
To see whether it is possible to target herpes with UV lamps?
Yes, you want to be able to trigger herpes in a targeted manner, and you also want to get by with just a few subjects, and you want to have results on the efficacy of your product in the shortest possible time – This is especially the goal in proof-of-concept studies with a small number of subjects/patients. In the mentioned study we could prove that the triggering of herpes by UV irradiation works well.
Are there any other benefits to this approach?
One thing that you can only do if you use UV light in a targeted and localized way to specifically induce herpes is the possibility of intra-individual control. By irradiating a very small area - about one square centimeter - with UV radiation on one side of the lips and inducing a cold sore there, we can reliably use the un-irradiated part of the lip as an intra-individual control region.
How well did that work?
The success rate was very good. 40% of the irradiated subjects developed a cold sore within two days and could be included in the study and treated. So that was excellent! There were practically no unwanted effects caused by the UV treatment. If patients just turned up sporadically at the trial site, like they would if you just recruit a lot of patients and wait for the cold sore to trigger, there are further limits. A study like ours, with special measurement methods would be impossible without plannable procedures.
What do you mean by special measurement methods?
Well, the second challenge, in addition to quickly and systematically obtaining a sufficient number of patients, was the investigational product itself: In the study we did not test an ordinary drug, but an occlusive patch, i.e. a plastic film that you stick on the lip. This was also coated with a drug, an antiviral local therapeutic agent. This means that all examinations had to be done through the plastic patch. Thus, you can only see the lesion indirectly through the plastic film. There are light scattering effects and so on, which makes it difficult to evaluate subjectively and even more to use measuring devices.
How did you approach the problem?
In principle, the question was which methods are suitable for seeing through these patches and measuring the healing process. In addition to the purely clinical assessment, three measurement methods were tested.
What methods were these?
The first method was temperature measurement by thermography. The skin affected by the cold sore has a higher temperature because of the inflammation. If the cold sore heals faster, for example due to treatment, then the temperature should also drop to normal faster.
The second parameter was Field Laser Perfusion Imaging (FLPI). With this, we measured the blood flow at the site of the lip where the herpes lesion originated. And there it's the same: high blood flow is a sign of inflammation. If you now make a blood flow curve over the treatment days, you can see how quickly the lesion heals.
The third method was clinical photography, where the idea was to measure the redness via image analytics.
These three methods were tried and it was not at all clear to us that any of them would work. So it was not only a question of seeing a therapeutic success, but it was also equally a question of which methods were suitable!
And were you successful? Do any of the measurement methods work through the patch?
Yes! As far as the results are concerned, we actually hoped to be able to measure the course of inflammation well, especially with thermography. However, this turned out to be more difficult than expected because we weren't measuring the surface of the lip skin, but the heat radiating from the surface of the patch. This means that we basically have an artifact; hence the thermography method wasn't very good in terms of results.
With the clinical photography, it was also quite difficult due to reflections from the surface of the patch.
Fortunately, we had a third method with FLPI that objectively measures blood flow. This has proven to be less affected by the patch, because it uses a long-wavelength laser that passes very well through the patch. This worked really well and we could actually measure the cold sore healing process and also show that the patch was effective.



So this type of design can be used in further efficacy studies of cold sore efficacy?
Absolutely!
Just out of interest: Was this actually a medical device or a drug?
Well, it is a patch, but it contains a viral static agent. It took about a year for the BfArM to decide that it was a medical device, because it was mainly the covering and the mechanical effect that played a role, and the drug only had a secondary effect, but that was not at all clear, it could also have turned out to be that it was essentially just a drug application. When you have new innovative systems, it is important to assess into which category they actually belong.
Is there actually something like a precedent system?
Of course, every BfArM decision on a certain topic can be cited again and the question would probably be decided much faster next time. But you also have to be very careful - the moment conditions turn out to be somewhat different, the materials for example, it is ultimately again necessary to have a case-by-case decision. Without consultation, you can never assume that you are always completely sure what category your innovative product falls into! But of course we can help with that, for example by doing a workshop with relevant experts for you. You can get a fairly reliable assessment or classification of the product in advance and will not be so surprised by the decision of BfArM!
And then nothing stands in the way of a compact clinical trial of a product for cold sores?
Exactly!
Related content
Herpes simplex proof of concept study – A case study
In this month's proderm Story we interviewed Stephan Bielfeldt, our Vice president Science and Consulting. He referenced a proof of concept study we conducted at proderm that highlights our capabilities when it comes to herpes simplex and we are excited to present you with a case study on this exploratory clinical study!
Research paper
We love nothing more than designing and conducting great studies together with our sponsors and publishing peer reviewed papers on our shared successes! Click the link to access the publication "A Randomized Exploratory Study to Investigate the Inflammatory Response During an Ultraviolet-Radiation-Induced Cold Sore Episode".
Testing approaches for cosmetic lip care products
The proderm story from April 2022 illustrates that herpes simplex can be caused by UV-radiation. However investigating lip care products goes far beyond the measurement of the sun protection factor. This article provides you with an overview of the most important aspects.