Investigation of baby care products
According to the EU Cosmetics Product Regulation, babies and children up to the age of three are particularly vulnerable consumers whose health is to be protected to a special degree. The skin of babies has specific physiological properties and is generally considered to be significantly more sensitive than the skin of an adult.
In this respect, there are good reasons to approach the subject of clinical testing of baby care products with the greatest possible care. At SGS proderm, we rely on professional scientific, sensitive and innovative examination methods as well as specialist medical care by pediatricians, dermatologists and allergologists as part of the examinations carried out at our company.
Study types
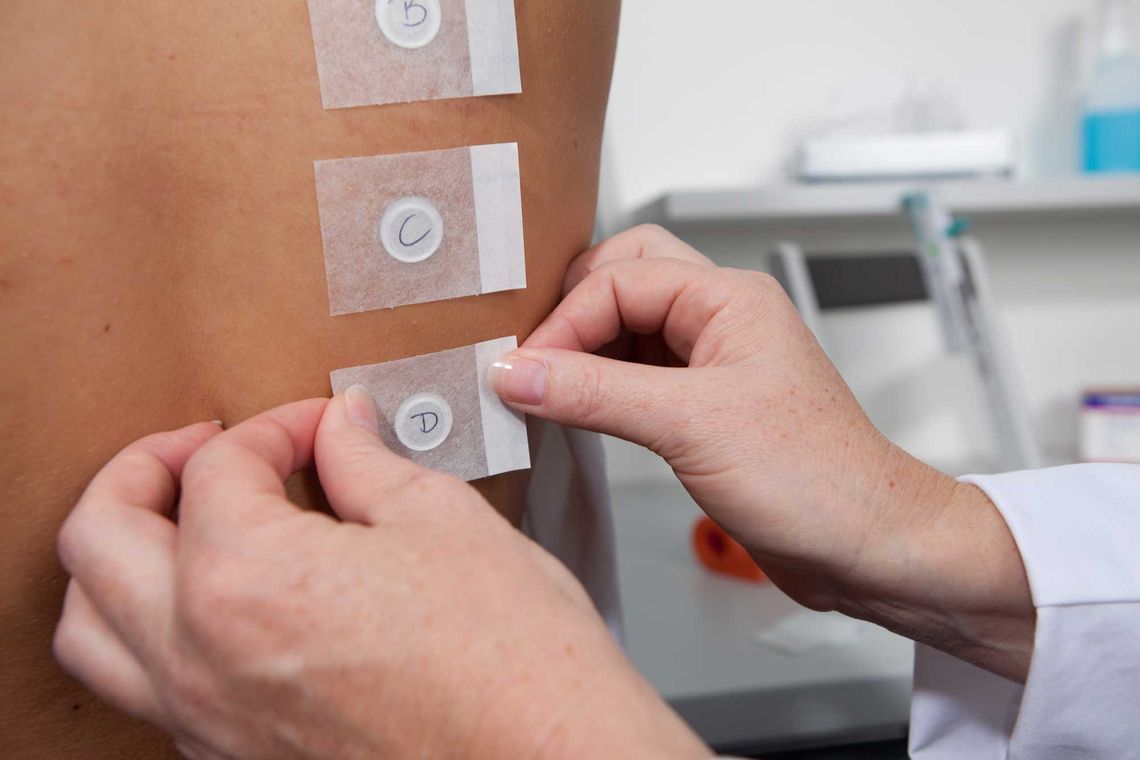
Product tolerability studies often involve deliberately excessive applications or minimally invasive treatments. These approaches cannot be reconciled with the special protective needs of babies, so the testing of compatibility of baby care products is done on adults as a first step.
It has proven successful to test the compatibility on adults with pre-damaged (or sensitive) skin and then to test the test product under pediatrician supervision in a baby use study. In the context of the tolerability of baby care products, the 'Eye Instillation No Tears Test' is also of particular relevance. Hair combability testing can also be of interest to manufacturers.
Testing of product efficacy is mostly focused on the topics of moisturization and barrier protection. Parameters such as transepidermal water loss (TEWL), skin moisturization and pH are frequently the subject of our investigations in this context. The thickness of the stratum corneum and the determination of Natural Moisturizing Factors (NMFs) by Raman spectroscopy provide further valuable insights into your product:
- Investigation of skin moisturization
- Measurement of skin barrier properties (TEWL)
- Determination of pH, stratum corneum thickness and NMFs